Fine-tune Enformer to perform binary classification on human snATAC-seq data#
In this tutorial, we learn how to fine-tune the Enformer model (https://www.nature.com/articles/s41592-021-01252-x) on the CATLAS single-nucleus ATAC-seq dataset from human tissues (http://catlas.org/humanenhancer/).
We will perform binary classification, in which the model learns to predict the probability that a given sequence is accessible in the different cell types of the dataset. For an example of regression modeling, see the next tutorial.
import anndata
import os
import importlib
import pandas as pd
import numpy as np
%matplotlib inline
Set experiment parameters#
experiment='tutorial_2'
if not os.path.exists(experiment):
os.makedirs(experiment)
Download data#
We download the CATlas ATAC-seq binary cell type x peak matrix from the gReLU model zoo. The original source of this data is https://decoder-genetics.wustl.edu/catlasv1/humanenhancer/data/cCRE_by_cell_type/. For more details, see https://decoder-genetics.wustl.edu/catlasv1/catlas_humanenhancer/#!/.
import grelu.resources
dataset_path = grelu.resources.download_dataset(repo_id="Genentech/tutorial-2-data")
Load data#
The download_dataset function returns the path to the downloaded h5ad file containing the binarized chromatin accessibility data for multiple human cell types. We now load this as an anndata object.
ad = anndata.read_h5ad(dataset_path)
ad
AnnData object with n_obs × n_vars = 222 × 1154611
obs: 'cell type'
var: 'chrom', 'start', 'end', 'Class'
This contains a binary matrix representing the accessibility of 1154611 CREs measured in 222 cell types. Let us look at the components of this object:
ad.var.head()
| chrom | start | end | Class | |
|---|---|---|---|---|
| 0 | chr1 | 9955 | 10355 | Promoter Proximal |
| 1 | chr1 | 29163 | 29563 | Promoter |
| 2 | chr1 | 79215 | 79615 | Distal |
| 3 | chr1 | 102755 | 103155 | Distal |
| 4 | chr1 | 115530 | 115930 | Distal |
ad.obs.head()
| cell type | |
|---|---|
| cell type | |
| Follicular | Follicular |
| Fibro General | Fibro General |
| Acinar | Acinar |
| T Lymphocyte 1 (CD8+) | T Lymphocyte 1 (CD8+) |
| T lymphocyte 2 (CD4+) | T lymphocyte 2 (CD4+) |
The contents of this anndata object are binary values (0 or 1). 1 indicates accessibility of the peak in the cell type.
ad.X[:5, :5].todense()
matrix([[1., 0., 0., 0., 0.],
[1., 0., 0., 0., 0.],
[1., 0., 0., 0., 0.],
[1., 0., 0., 0., 0.],
[1., 0., 0., 0., 0.]], dtype=float32)
Filter peaks#
We will perform filtering of this dataset using the grelu.data.preprocess module.
First, we filter peaks within autosomes (chromosomes 1-22) or chromsomes X/Y. You can also supply autosomes or autosomesX to further restrict the chromosomes.
import grelu.data.preprocess
ad = grelu.data.preprocess.filter_chromosomes(ad, 'autosomesXY')
Keeping 1154464 intervals
Next, we drop peaks overlapping with the ENCODE blacklist regions for the hg38 genome.
ad = grelu.data.preprocess.filter_blacklist(ad, genome='hg38')
Keeping 1149915 intervals
Visualize data#
Next, we can plot the distribution of the data in various ways.
import grelu.visualize
%matplotlib inline
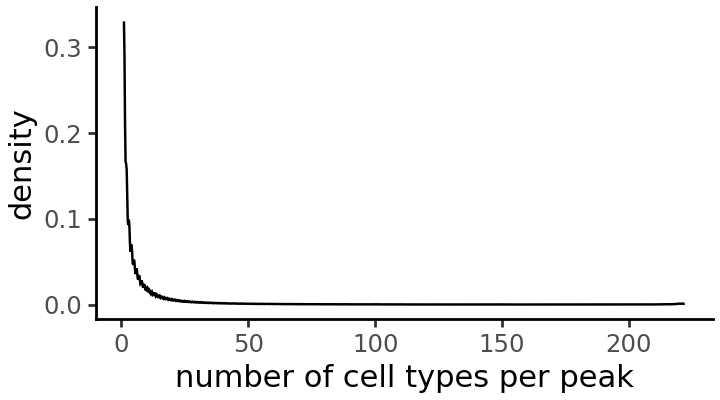
In how many accessible cell types is each peak accessible?
cell_types_per_peak = np.array(np.sum(ad.X > 0, axis=0))
grelu.visualize.plot_distribution(
cell_types_per_peak,
title='number of cell types per peak',
method='density', # Alternative: histogram
figsize=(3.6, 2), # width, height
)
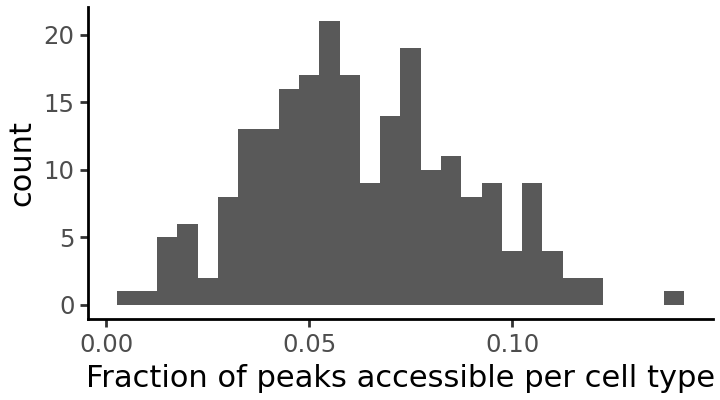
How many peaks are accessible in each cell type?
fraction_accessible_per_cell_type = np.array(np.mean(ad.X > 0, axis=1))
grelu.visualize.plot_distribution(
fraction_accessible_per_cell_type,
title='Fraction of peaks accessible per cell type',
method='histogram', # alternative: density
binwidth=0.005,
figsize=(3.6, 2), # width, height
)
It seems that some cell types have very few accessible peaks. We will drop these cell types from the dataset.
print(ad.shape)
ad = ad[ad.X.mean(axis=1) > .03, :]
print(ad.shape)
(222, 1149915)
(203, 1149915)
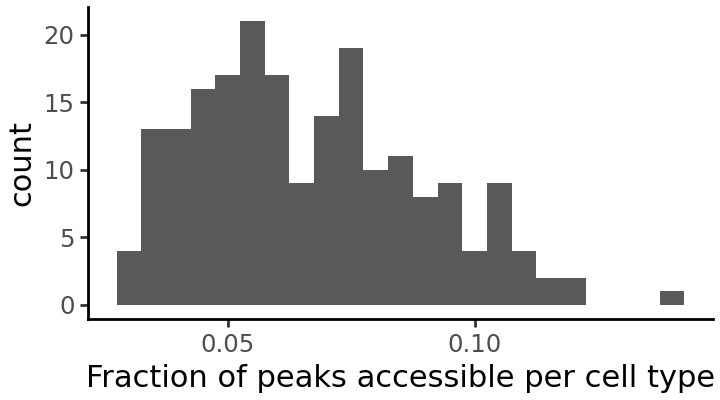
We can plot the distribution once again to see the effect of this filtering:
Resize peaks#
Finally, since the ATAC-seq peaks can have different lengths, we resize all of them to a constant length in order to train the model. Here, we take 200 bp. We can use the resize function in grelu.sequence.utils, which contains functions to manipulate DNA sequences.
import grelu.sequence.utils
seq_len = 200
ad.var = grelu.sequence.utils.resize(ad.var, seq_len)
ad.var.head(3)
| chrom | start | end | Class | |
|---|---|---|---|---|
| 27 | chr1 | 794997 | 795197 | Distal |
| 28 | chr1 | 804833 | 805033 | Promoter Proximal |
| 29 | chr1 | 816263 | 816463 | Promoter Proximal |
Split data#
We will split the peaks by chromosome to create separate sets for training, validation and testing.
train_chroms='autosomes'
val_chroms=['chr10']
test_chroms=['chr11']
ad_train, ad_val, ad_test = grelu.data.preprocess.split(
ad,
train_chroms=train_chroms, val_chroms=val_chroms, test_chroms=test_chroms,
)
Selecting training samples
Keeping 1004076 intervals
Selecting validation samples
Keeping 56911 intervals
Selecting test samples
Keeping 56366 intervals
Final sizes: train: (203, 1004076), val: (203, 56911), test: (203, 56366)
Make labeled sequence datasets#
grelu.data.dataset contains PyTorch Dataset classes that can load and process genomic data for training a deep learning model. Here, we use the AnnDataSeqDataset class which loads data from an AnnData object. Other available dataset classes include DFSeqDataset and BigWigSeqDataset.
We first make the training dataset. To increase model robustness we use several forms of data augmentation here: rc=True (reverse complementing the input sequence), and max_seq_shift=1 (shifting the coordinates of the input sequence by upto 1 bp in either direction; also known as jitter). We use augmentation_mode="random" which means that at each iteration, the model sees a randomly selected augmented version for each sequence.
import grelu.data.dataset
train_dataset = grelu.data.dataset.AnnDataSeqDataset(
ad_train.copy(),
genome='hg38',
rc=True, # reverse complement
max_seq_shift=1, # Shift the sequence
augment_mode="random", # Randomly select which augmentations to apply
)
We do not apply any augmentations to the validation and test datasets (although it is possible to do so).
val_dataset = grelu.data.dataset.AnnDataSeqDataset(ad_val.copy(), genome='hg38')
test_dataset = grelu.data.dataset.AnnDataSeqDataset(ad_test.copy(), genome='hg38')
Build the enformer model#
gReLU contains many model architectures. One of these is a class called EnformerPretrainedModel. This class creates a model identical to the published Enformer model and initialized with the trained weights, but where you can change the number of transformer layers and the output head.
Models are created using the grelu.lightning module. In order to instantiate a model, we need to specify model_params (parameters for the model architecture) and train_params (parameters for training).
model_params = {
'model_type':'EnformerPretrainedModel', # Type of model
'n_tasks': ad.shape[0], # Number of cell types to predict
'crop_len':0, # No cropping of the model output
'n_transformers': 1, # Number of transformer layers; the published Enformer model has 11
}
train_params = {
'task':'binary', # binary classification
'lr':1e-4, # learning rate
'logger': 'csv', # Logs will be written to a CSV file
'batch_size': 1024,
'num_workers': 8,
'devices': 0, # GPU index
'save_dir': experiment,
'optimizer': 'adam',
'max_epochs': 10,
'checkpoint': True, # Save checkpoints
}
import grelu.lightning
model = grelu.lightning.LightningModel(model_params=model_params, train_params=train_params)
Train the model#
We train the model on the new training dataset using the train_on_dataset method. Note that here, we update the weights of the entire model during training. If you want to hold the enformer weights fixed and only learn a linear layer from the enformer embedding to the outputs, see the tune_on_dataset method.
# See the 'tutorial_2' folder for logs
trainer = model.train_on_dataset(
train_dataset=train_dataset,
val_dataset=val_dataset,
)
/gpfs/scratchfs01/site/u/lala8/conda/envs/grelu-test/lib/python3.10/site-packages/rich/live.py:260: UserWarning: install "ipywidgets" for Jupyter support
┏━━━━━━━━━━━━━━━━━━━━━━━━━━━┳━━━━━━━━━━━━━━━━━━━━━━━━━━━┓ ┃ Validate metric ┃ DataLoader 0 ┃ ┡━━━━━━━━━━━━━━━━━━━━━━━━━━━╇━━━━━━━━━━━━━━━━━━━━━━━━━━━┩ │ val_accuracy │ 0.49347996711730957 │ │ val_auroc │ 0.49767163395881653 │ │ val_avgprec │ 0.10673108696937561 │ │ val_best_f1 │ 0.17083537578582764 │ │ val_loss │ 0.6943522095680237 │ └───────────────────────────┴───────────────────────────┘
┏━━━┳━━━━━━━━━━━━━━┳━━━━━━━━━━━━━━━━━━━━━━━━━┳━━━━━━━━┳━━━━━━━┳━━━━━━━┓ ┃ ┃ Name ┃ Type ┃ Params ┃ Mode ┃ FLOPs ┃ ┡━━━╇━━━━━━━━━━━━━━╇━━━━━━━━━━━━━━━━━━━━━━━━━╇━━━━━━━━╇━━━━━━━╇━━━━━━━┩ │ 0 │ model │ EnformerPretrainedModel │ 72.1 M │ train │ 0 │ │ 1 │ loss │ BCEWithLogitsLoss │ 0 │ train │ 0 │ │ 2 │ val_metrics │ MetricCollection │ 0 │ train │ 0 │ │ 3 │ test_metrics │ MetricCollection │ 0 │ train │ 0 │ │ 4 │ transform │ Identity │ 0 │ train │ 0 │ └───┴──────────────┴─────────────────────────┴────────┴───────┴───────┘
Trainable params: 72.1 M Non-trainable params: 0 Total params: 72.1 M Total estimated model params size (MB): 288 Modules in train mode: 240 Modules in eval mode: 0 Total FLOPs: 0
/gpfs/scratchfs01/site/u/lala8/conda/envs/grelu-test/lib/python3.10/site-packages/rich/live.py:260: UserWarning: install "ipywidgets" for Jupyter support
/gpfs/scratchfs01/site/u/lala8/conda/envs/grelu-test/lib/python3.10/site-packages/rich/live.py:260: UserWarning: install "ipywidgets" for Jupyter support
Load best model from checkpoint#
During training, the performance of the model on the validation set is checked after each epoch. We will load the version of the model that had the best validation set performance.
best_checkpoint = trainer.checkpoint_callback.best_model_path
print(best_checkpoint)
model = grelu.lightning.LightningModel.load_from_checkpoint(best_checkpoint)
tutorial_2/2026_03_03_15_54/version_0/checkpoints/epoch=7-step=7848.ckpt
Calculate performance metrics on the test set#
We calculate global performance metrics using the test_on_dataset method.
test_metrics = model.test_on_dataset(
test_dataset,
devices=0,
num_workers=8,
batch_size=1024,
)
┏━━━━━━━━━━━━━━━━━━━━━━━━━━━┳━━━━━━━━━━━━━━━━━━━━━━━━━━━┓ ┃ Test metric ┃ DataLoader 0 ┃ ┡━━━━━━━━━━━━━━━━━━━━━━━━━━━╇━━━━━━━━━━━━━━━━━━━━━━━━━━━┩ │ test_accuracy │ 0.9458667039871216 │ │ test_auroc │ 0.9044811725616455 │ │ test_avgprec │ 0.6075653433799744 │ │ test_best_f1 │ 0.5681172013282776 │ │ test_loss │ 0.15379782021045685 │ └───────────────────────────┴───────────────────────────┘
test_metrics is a dataframe containing metrics for each model task.
test_metrics.head()
| test_accuracy | test_auroc | test_avgprec | test_best_f1 | |
|---|---|---|---|---|
| Follicular | 0.909236 | 0.878971 | 0.628441 | 0.578610 |
| Fibro General | 0.916510 | 0.883105 | 0.625623 | 0.575389 |
| Acinar | 0.944186 | 0.913036 | 0.650671 | 0.602352 |
| T Lymphocyte 1 (CD8+) | 0.962034 | 0.930548 | 0.635034 | 0.589981 |
| T lymphocyte 2 (CD4+) | 0.967108 | 0.939110 | 0.618744 | 0.573214 |
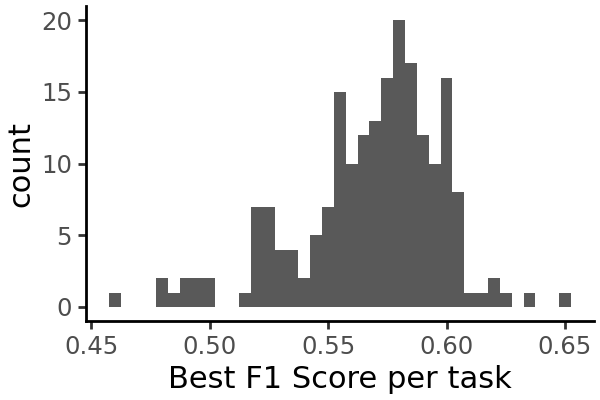
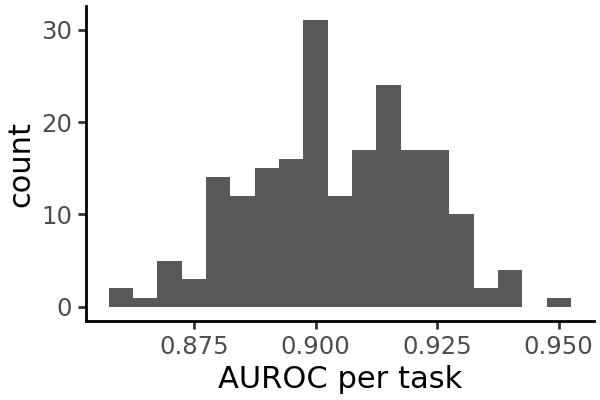
Visualize performance metrics#
We can plot the distribution of each metric across all cell types:
grelu.visualize.plot_distribution(
test_metrics.test_best_f1,
method='histogram',
title='Best F1 Score per task',
binwidth=0.005,
figsize=(3,2),
)
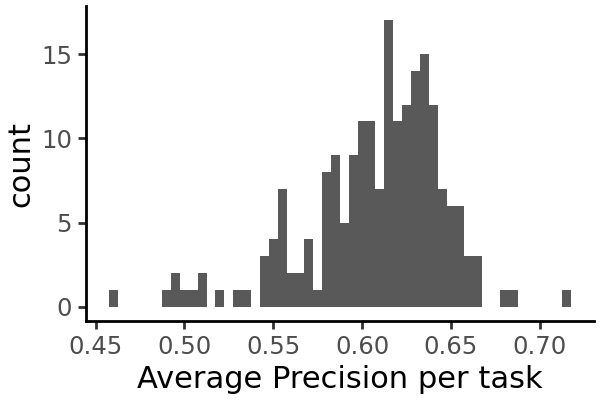
grelu.visualize.plot_distribution(
test_metrics.test_avgprec,
method='histogram',
title='Average Precision per task',
binwidth=0.005,
figsize=(3,2),
)
Run inference on the test set#
Instead of overall metrics, we can also get the individual predictions for each test set example.
probs = model.predict_on_dataset(
test_dataset,
devices=0,
num_workers=8,
batch_size=1024,
return_df=True # Return the output as a pandas dataframe
)
probs.head()
| Follicular | Fibro General | Acinar | T Lymphocyte 1 (CD8+) | T lymphocyte 2 (CD4+) | Natural Killer T | Naive T | Fibro Epithelial | Cardiac Pericyte 1 | Pericyte General 1 | ... | Fetal Cardiac Fibroblast | Fetal Fibro General 2 | Fetal Fibro Muscle 1 | Fetal Fibro General 3 | Fetal Mesangial 2 | Fetal Stellate | Fetal Alveolar Epithelial 1 | Fetal Cilliated | Fetal Excitatory Neuron 1 | Fetal Excitatory Neuron 2 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 0.037320 | 0.059341 | 0.019623 | 0.057960 | 0.022940 | 0.037132 | 0.024256 | 0.060020 | 0.036380 | 0.044111 | ... | 0.096488 | 0.084068 | 0.098517 | 0.111082 | 0.123722 | 0.055901 | 0.168764 | 0.117609 | 0.134788 | 0.044917 |
| 1 | 0.045293 | 0.040371 | 0.026782 | 0.025121 | 0.012357 | 0.010523 | 0.011736 | 0.027949 | 0.032551 | 0.026512 | ... | 0.084573 | 0.056361 | 0.049276 | 0.085011 | 0.076023 | 0.030324 | 0.179506 | 0.081972 | 0.063167 | 0.018129 |
| 2 | 0.076708 | 0.127368 | 0.057874 | 0.060736 | 0.036988 | 0.032808 | 0.036318 | 0.105995 | 0.112944 | 0.101309 | ... | 0.183218 | 0.162204 | 0.136362 | 0.171209 | 0.238024 | 0.089201 | 0.274355 | 0.169164 | 0.204561 | 0.066065 |
| 3 | 0.071490 | 0.027930 | 0.013738 | 0.020133 | 0.012074 | 0.013508 | 0.009373 | 0.023522 | 0.023282 | 0.020539 | ... | 0.033375 | 0.021606 | 0.019337 | 0.032877 | 0.030444 | 0.012405 | 0.072734 | 0.055420 | 0.034482 | 0.014244 |
| 4 | 0.012588 | 0.018428 | 0.010383 | 0.121269 | 0.102168 | 0.110026 | 0.067189 | 0.007175 | 0.014089 | 0.004672 | ... | 0.009173 | 0.005470 | 0.005460 | 0.012376 | 0.007533 | 0.003261 | 0.038494 | 0.018055 | 0.011567 | 0.006286 |
5 rows × 203 columns
Since this is a binary classification model, the output takes the form of probabilities ranging from 0 to 1. We interpret these as the predicted probabilities of the element being accessible in each cell type.
Plot additional visualizations of the test set predictions#
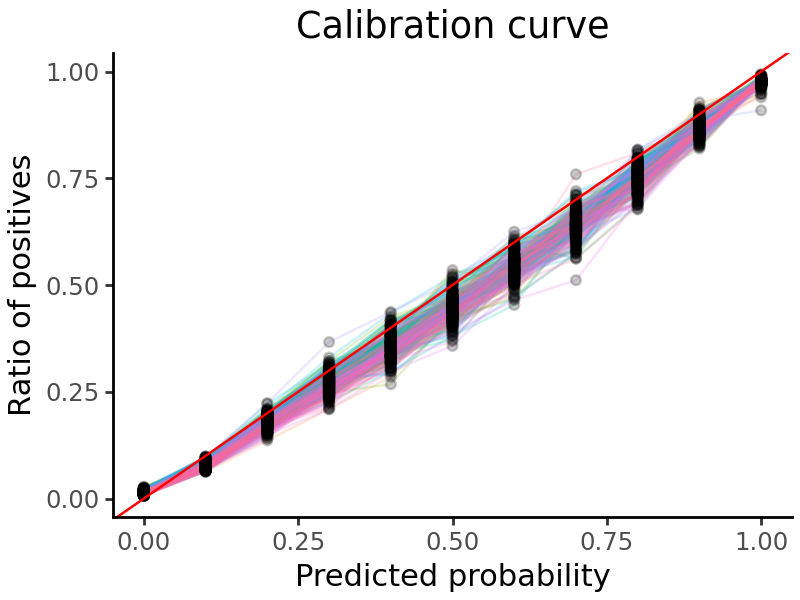
We can plot a calibration curve for all the tasks. This shows us the fraction of true positive examples, for different levels of model-predicted probability.
grelu.visualize.plot_calibration_curve(
probs, labels=test_dataset.labels, aggregate=False, show_legend=False
)
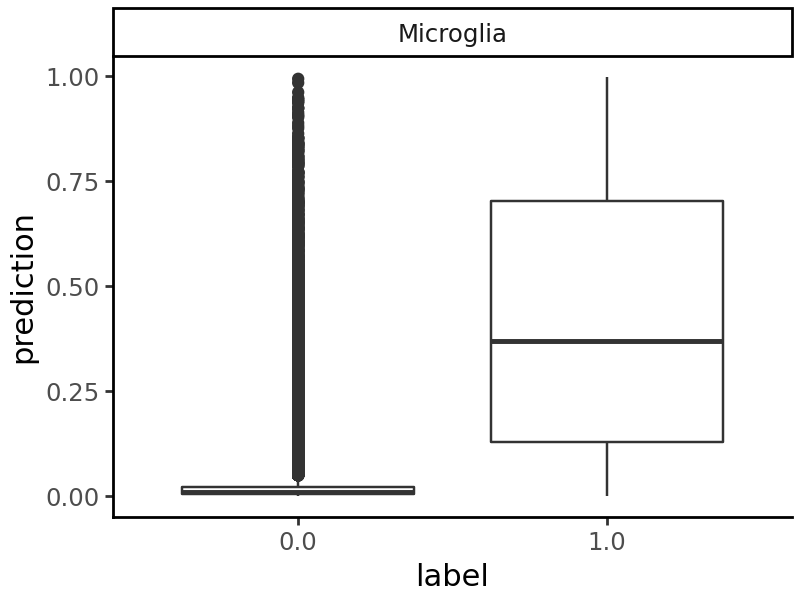
We can also pick any cell type and compare the predictions on accessible and non-accessible elements.
Interpret model predictions (for microglia) using TF-modisco#
Suppose we want to focus specifically on Microglia. We can create a transform - a class that takes in the model’s prediction and returns a function of the prediction, e.g. the prediction for only a subset of cell types that we are interested in. Then, all subsquent analyses that we do will be based only on this subset of the full prediction matrix.
Transforms can also compute more complicated functions of the model’s output - for example, the Specificity class of transforms take in the full set of predictions produced by the model and compute the predicted specificity of each sequence to a particular cell type or otuput track.
In this example, we use the Specificity transform to analyze the predicted cell type specificity of sequences in microglia - i.e. the predicted accessibility in microglia minus the mean predicted accessibility in other cell types. See the Aggregate class if you want to just subset the predictions to microglia.
from grelu.transforms.prediction_transforms import Specificity
microglia_scorer = Specificity(
on_tasks = ["Microglia"],
model = model,
compare_func='subtract' # Subtract the prediction in other cell types from the prediction in microglia
)
The simplest way to use a transform is to apply it directly to the model’s predictions using the .compute() method. Here, we apply this microglia specificity transform to the test set predictions:
# Transforms expect a 3-D input - add an axis 2 to the predictions.
mcg_specificity = microglia_scorer.compute(np.expand_dims(probs, 2)).squeeze()
# This is the predicted specificity for each test sequence in microglia
mcg_specificity[:5]
array([-0.02669976, -0.0286902 , -0.06530956, -0.01327392, 0.03015377],
dtype=float32)
We can now select the 250 test set peaks with the highest predicted specificity in microglia:
mcg_peaks = ad.var.iloc[np.argsort(mcg_specificity)].tail(250)
mcg_peaks
| chrom | start | end | Class | |
|---|---|---|---|---|
| 48604 | chr1 | 110228431 | 110228631 | Distal |
| 9045 | chr1 | 17426393 | 17426593 | Distal |
| 56231 | chr1 | 153465814 | 153466014 | Distal |
| 22304 | chr1 | 43446480 | 43446680 | Promoter Proximal |
| 17265 | chr1 | 32701425 | 32701625 | Promoter Proximal |
| ... | ... | ... | ... | ... |
| 54009 | chr1 | 146987608 | 146987808 | Distal |
| 18729 | chr1 | 36233365 | 36233565 | Distal |
| 52592 | chr1 | 118552721 | 118552921 | Distal |
| 12489 | chr1 | 23645409 | 23645609 | Distal |
| 22411 | chr1 | 43595659 | 43595859 | Distal |
250 rows × 4 columns
But transforms are more powerful than this - they allow you to do any downstream task, e.g. interpretation, variant effect prediction and design, with respect to the computed quantity.
Here, we will run TF-Modisco on these selected peaks. TF-Modisco identifies motifs that consistently contribute to the model’s output. Since we are using the microglia_scorer filter to compute predicted specificity to microglia, we will only get motifs that contribute to increased or decreased specificity. We also use TOMTOM to match the TF-Modisco motifs to a set of reference motifs. Here, we use the HOCOMOCO v12 motif set (https://hocomoco12.autosome.org/).
See the subsequent tutorials for more examples of how to use transforms.
%%time
import grelu.interpret.modisco
grelu.interpret.modisco.run_modisco(
model,
seqs=mcg_peaks,
genome="hg38",
prediction_transform=microglia_scorer, # Attributions will be calculated with respect to this output
meme_file="hocomoco_v13", # We will compare the Modisco CWMs to HOCOMOCO motifs
method="saliency", # Base-level attribution scores will be calculated using saliency. You can also use ISM here.
correct_grad=True, # Gradient correction; only applied with saliency
out_dir=experiment,
batch_size=256,
devices=0,
num_workers=32,
seed=0,
# Here, we are demonstrating this function on a small number of peaks,
# So we set these modisco parameters to a low value. In real runs, we suggest
# using larger or default values.
min_metacluster_size=6,
final_min_cluster_size=6,
)
Getting attributions
Running modisco
4 positive and 5 negative patterns were found.
Writing modisco output
Creating sequence logos
Creating html report
Running TOMTOM
CPU times: user 1min 46s, sys: 1.74 s, total: 1min 47s
Wall time: 1min 24s
Load TOMTOM output for modisco motifs#
The full output of TF-Modisco and TOMTOM can be found in the experiment folder. Here, we read the output of TOMTOM and list the significant TOMTOM matches, i.e. known TF motifs that are similar to those found by TF-MoDISco.
tomtom_file = os.path.join(experiment, 'tomtom.csv')
tomtom = pd.read_csv(tomtom_file, index_col=0)
tomtom[tomtom['q-value'] < 0.001] # Display most significant matches
| Query_ID | Target_ID | Optimal_offset | p-value | E-value | q-value | Overlap | Query_consensus | Target_consensus | Orientation | |
|---|---|---|---|---|---|---|---|---|---|---|
| 175 | pos_pattern_0 | EHF.H13CORE.0.P.B | 2.0 | 8.607895e-08 | 0.000139 | 0.000195 | 13.0 | CACTTCCTCCTTG | GAACCAGGAAGTGGG | - |
| 182 | pos_pattern_0 | ELF4.H13CORE.1.M.B | 1.0 | 1.090203e-06 | 0.001756 | 0.000930 | 13.0 | CACTTCCTCCTTG | GGAAACAGGAAGTAA | - |
| 183 | pos_pattern_0 | ELF5.H13CORE.0.PSM.A | 1.0 | 5.438433e-08 | 0.000088 | 0.000195 | 13.0 | CACTTCCTCCTTG | AGGAAGGAGGAAGTAA | - |
| 192 | pos_pattern_0 | ERF.H13CORE.0.PS.A | 0.0 | 1.100122e-07 | 0.000177 | 0.000195 | 11.0 | CACTTCCTCCTTG | AACAGGAAGTG | - |
| 207 | pos_pattern_0 | ETS2.H13CORE.1.P.B | 1.0 | 7.843389e-07 | 0.001264 | 0.000758 | 11.0 | CACTTCCTCCTTG | GACCGGAAGTGG | - |
| 218 | pos_pattern_0 | ETV6.H13CORE.1.P.B | 1.0 | 8.021184e-09 | 0.000013 | 0.000058 | 11.0 | CACTTCCTCCTTG | AAGAGGAAGTGG | - |
| 997 | pos_pattern_0 | SPI1.H13CORE.0.P.B | 1.0 | 4.753351e-09 | 0.000008 | 0.000058 | 13.0 | CACTTCCTCCTTG | AAAAGAGGAAGTGA | - |
| 998 | pos_pattern_0 | SPI1.H13CORE.1.S.B | 1.0 | 9.519393e-08 | 0.000153 | 0.000195 | 11.0 | CACTTCCTCCTTG | AAGGGGAAGTAG | - |
| 999 | pos_pattern_0 | SPIB.H13CORE.0.P.B | 4.0 | 3.030316e-08 | 0.000049 | 0.000146 | 12.0 | CACTTCCTCCTTG | AAAGAGGAAGTGAAAG | - |
| 1001 | pos_pattern_0 | SPIB.H13CORE.2.SM.B | 1.0 | 9.557814e-07 | 0.001540 | 0.000866 | 13.0 | CACTTCCTCCTTG | GGGAATGAGGAAGTAG | - |
| 2101 | pos_pattern_1 | KLF14.H13CORE.1.P.C | -14.0 | 7.038565e-07 | 0.001134 | 0.000729 | 23.0 | AGGTCAACCATACGCCCACTCCCGTCACTCCCCCGAGG | GAGGGGGCGGGGCCGGGGGGGGG | - |
| 2971 | pos_pattern_1 | ZN557.H13CORE.0.P.C | -2.0 | 6.183836e-07 | 0.000996 | 0.000690 | 25.0 | AGGTCAACCATACGCCCACTCCCGTCACTCCCCCGAGG | GAACCTGGAAGTGGATATTTGTGGG | - |
| 3731 | pos_pattern_2 | KMT2A.H13CORE.0.P.B | -6.0 | 1.209336e-07 | 0.000195 | 0.000195 | 23.0 | GGCCTCACGAGGCCCAGGCGCCGCCGCGCGGGCGCCAGCGGCCCCAC | CCCCGCCGCCGCCGCCGCCGCCC | + |
| 6474 | neg_pattern_0 | ATF3.H13CORE.0.P.B | 0.0 | 3.897465e-07 | 0.000628 | 0.000514 | 7.0 | TGAGTCA | GGATGACTCA | - |
| 6986 | neg_pattern_0 | MAFA.H13CORE.1.M.C | 4.0 | 2.608469e-07 | 0.000420 | 0.000378 | 7.0 | TGAGTCA | AAAATTGCTGACTCAGCAA | - |
| 6993 | neg_pattern_0 | MAFK.H13CORE.0.PS.A | 3.0 | 1.169238e-06 | 0.001884 | 0.000942 | 7.0 | TGAGTCA | AAAATTGCTGACTCAGCA | - |
| 7076 | neg_pattern_0 | NF2L2.H13CORE.0.P.B | 2.0 | 7.294213e-08 | 0.000118 | 0.000195 | 7.0 | TGAGTCA | GCTGAGTCATGCTGA | + |
| 11928 | neg_pattern_3 | NFIA.H13CORE.0.P.B | -20.0 | 5.547534e-07 | 0.000894 | 0.000670 | 8.0 | ATCTGAGACTCTGGGGAAGCCTGCCAGGACCTCATCC | CTTGGCAG | - |